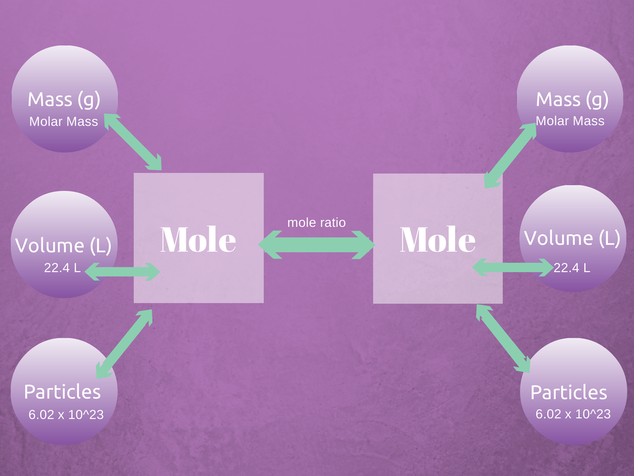
Balancing Equations
A equation will be balanced when the coefficient numbers are proportional and they represent the number of moles or particles in an equation.
1. Moles
The coefficient tells us how much moles are in a substance
2. Mass
The Law of Conservation of Mass says that mass is neither created or destroyed; it is always conserved.
1. Moles
The coefficient tells us how much moles are in a substance
2. Mass
The Law of Conservation of Mass says that mass is neither created or destroyed; it is always conserved.
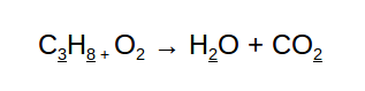
In this equations there are
(Reactant Side) (Product Side)
3 Carbon atoms 1 Carbon atom
8 Hydrogen atoms 2 Hydrogen atoms
2 Oxygen atoms 3 Oxygen atoms
(Reactant Side) (Product Side)
3 Carbon atoms 1 Carbon atom
8 Hydrogen atoms 2 Hydrogen atoms
2 Oxygen atoms 3 Oxygen atoms
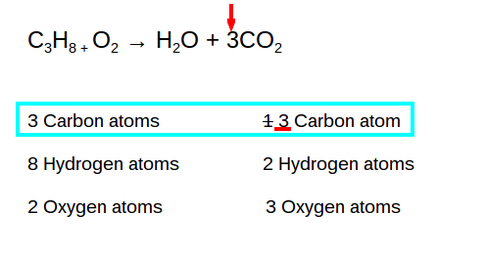
There was 1 Carbon atom on the product side and 3 Carbon atoms on the reactant side. In order to be balanced, there had to be 3 carbon atoms on each side so the 1 turned into a 3.
*You can change the coefficients but never change the subscripts
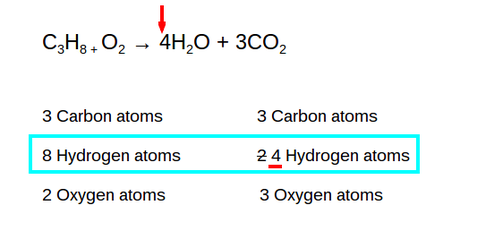
There are 8 Hydrogen atoms on the reactant side so there has to be 8 on the product side. There are 2 Hydrogen atoms on the product side. You have to put a 4 as the coefficient so that it will equal 8. So the 2 turned into a 4. (4x2=8)

You now have 10 Oxygen atoms on the product side. The 4 in front of H2O applies to all so you have 4 Oxygen atoms. Then the 3 in front of the CO2 applies to all so you have 6 oxygen atoms. Altogether, you have 10.

Since there are 10 Oxygen atoms on the product side, you need to have 10 on the reactant side. You have 2 oxygen atoms on the reactant side so in order to get it to 10, you have to multiply by 5.
mole ratio
*Treat coefficients in a balanced equation as mole ratios
If there is no number in front of a substance then it is considered a 1
We'll use the same equation as above.
C3H8 + 5O2 -> 4H2O + 3CO2
Ratio of C3H8 to O2; 1C3H8: 5O2 Ratio of C3H8 to 3CO2; 1C3H8: 3CO2
Ratio of O2 to 4H20; 5O2: 4H20 Ratio of 4H20 to 3CO2; 4H20: 3CO2
If there is no number in front of a substance then it is considered a 1
We'll use the same equation as above.
C3H8 + 5O2 -> 4H2O + 3CO2
Ratio of C3H8 to O2; 1C3H8: 5O2 Ratio of C3H8 to 3CO2; 1C3H8: 3CO2
Ratio of O2 to 4H20; 5O2: 4H20 Ratio of 4H20 to 3CO2; 4H20: 3CO2
Juan, Emily. "Stoichiometry Tutorial (Chemistry)." YouTube. YouTube, 12 June 2013. Web. 23 May 2014.